Olefination represents the reaction that introduces carbon-carbon double bonds into a chemical molecule to form the olefins (C=C, also known as alkene). Given the significant effect of C=C on enhancing the pharmacological activity and altering several physicochemical properties for many compounds, olefin products are, therefore, widely utilized in the fields of chemical research, pharmaceutical industry and agriculture.
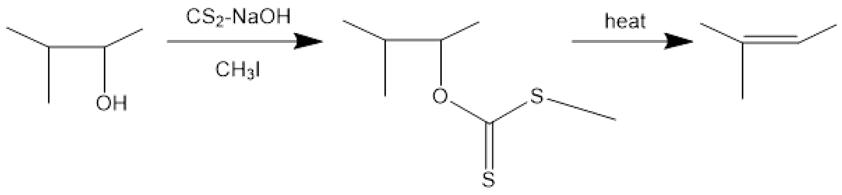 Fig 1. Synthesis of 2-methyl-2-butene
Fig 1. Synthesis of 2-methyl-2-butene
Cope Elimination, also can be termed as cope reaction, represents an elimination reaction of thermal decomposition of amine oxides under a high temperature condition (150-200°C) to produce hydroxylamines and olefins. The underlying mechanism for such elimination reaction mainly involves an intramolecular 5-membered cyclic transition state. Cope reaction is widely applied for olefin synthesis and removal of nitrogen in compounds for its highly efficient and accurate.
 Fig 2. Synthesis of Propylene
Fig 2. Synthesis of Propylene
Wittig reaction, discovered in 1954 by Georg Wittig, is the most widely used for the conversion of carbonyl compounds including aldehydes, ketones, esters, amides and lactams to the corresponding olefin derivatives. It is of great value for the preparation of compounds that interest to the chemical, agrochemical and pharmaceutical industries. However, the sterically hindered ketones in the structure of compounds may lead to a low rate of reaction and a poor yield. In this case, the Horner–Wadsworth–Emmons (HWE) reaction (using phosphonate esters) is recommended as an alternative approach.
 Fig 3. Synthesis of Isobutylene
Fig 3. Synthesis of Isobutylene
As an improvement of the Wittig reaction, Horner-Wadsworth-Emmons (HWE) reaction is applied in organic chemistry with aldehydes (or ketones) to produce predominantly alkenes. During the process of such reaction, a stable phosphonate carbanion instead of phosphorus ylide is used to react with aldehydes and ketones to produce olefins. An electron-withdrawing group (EWG) is required to be attached to the α-carbon of the phosphonate generally participating in the reaction, so that the four-membered ring intermediate in the reaction is eliminated to generate olefins.
 Fig 4. Synthesis of Ethylester
Fig 4. Synthesis of Ethylester
BOC Sciences has great capabilities and expertise to assist you with development, piloting or manufacture of your olefination process. Using our unique range of equipment, we can work with customers to develop and optimize the most efficient and economic olefination strategy. Additionally, our top-notch analysis and purification platforms enable our customers can be offered with the highly pure products to meet the requirements of impurities, by-product and intermediate controlling.
We offer olefination services to construct carbon–carbon double bonds, enabling molecular complexity and diversity.
Submit your inquiry to request a custom solution.
References
If you have any questions or encounter issues on this page, please don't hesitate to reach out. Our support team is ready to assist you.