Steroids are natural chemicals widely found in nature. It is a kind of lipid compound and characterized by a four ring parent nucleus of all steroids derived from the acetyl coenzyme A synthesis pathway. Different steroids are only diverse in their functional groups that attached to their rings, and their basic structure is the cyclopentane polyhydric phenanthrene. Commonly used steroids include phytosterols, bile acids, C21 steroids, insect metamorphic hormones, cardiac glycosides, steroidal saponins, steroid alkaloids, and toad venom.
Steroids are one of the most important substances in biological systems, and steroid hormones are combined with their receptor proteins to produce physiological responses, causing changes in gene transcription and cell function. Given the significant importance of steroids, the synthesis and application of steroids is attracting more and more attention in pharmaceutical industry.
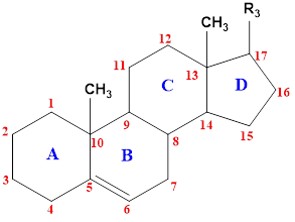 Fig 1. The general structure of steroids
Fig 1. The general structure of steroids
Steroids are a class of biomolecules that exist widely and have important functions in organisms. Its special structure makes the compounds have lipophilicity, membrane affinity and specific binding with low density lipoprotein. Using these characteristics, we design and synthesize steroid conjugates of various drug molecules, which can increase the liposoluble and improve the transmembrane permeability of drug molecules.
3-oxo-△4'6 Steroids are important intermediates in the synthesis of steroid drugs. The synthesis of this compound is of great significance. The synthetic methods of 3-oxo-△4'6 Steroids are manganese dioxide dehydrogenation method, palladium carbon dehydrogenation method and so on.
Steroid alkaloid Cortistatin compounds are another new type of angiogenesis inhibitor after the marine amino sterol compound squalamine. Since their unique chemical structure and significant biological activity have aroused great interest in organic synthesis chemists, the synthesis of Cortistatin A in more than two years has been made. It was completed by semi synthesis and total synthesis, and many strategies for constructing Cortistatin class B marine natural products were also reported.
 Fig 2. Synthesis Route Design of Cortistatin A (Yong Shi et al. 2010)
Fig 2. Synthesis Route Design of Cortistatin A (Yong Shi et al. 2010)
Hydroxylated steroids play an important role in improving drug activity, cytotoxicity and metabolism of exogenous compounds. However, as an important pharmaceutical intermediate, hydroxysteroids have a large market demand, complicated extraction processes, and difficult chemical synthesis, which is not conducive to the development of innovative drugs. We hope to solve these problems through biocatalysis (here refers to Enzymology). Enzyme catalyzed methods have attracted more and more attention because of the advantages of short production cycle, high yield, mild reaction conditions, high regional selectivity and stereoselectivity.
BOC Sciences is proud to provide you high-quality chemical synthesis service. Our experts and chemists have been developing and implementing a wide variety of drug synthesis pathways. We have a dedicated custom synthesis team, as well as starting materials and intermediates for various synthetic compounds. In addition, various techniques (NMR, LCMS, GCMS, HPLC, IR, and X-ray) are used to analyze the synthesis of the completed compound, and synthesis is in a short turnaround time.
We deliver steroid synthesis with reproducible purity and structure, ensuring dependable results for biochemical and industrial uses.
Submit your inquiry to request a custom solution.
References
If you have any questions or encounter issues on this page, please don't hesitate to reach out. Our support team is ready to assist you.